Ciprofloxacin; Dexamethasone Patent Expiration
Ciprofloxacin; Dexamethasone is Used for treating ear infections. It was first introduced by Sandoz Inc
Ciprofloxacin; Dexamethasone Patents
Given below is the list of patents protecting Ciprofloxacin; Dexamethasone, along with the drug name that holds that patent and the company name owning that drug.
| Drug Used in | Drug Patent Number | Drug Patent Title | Drug Patent Expiry | Drug Owner |
|---|---|---|---|---|
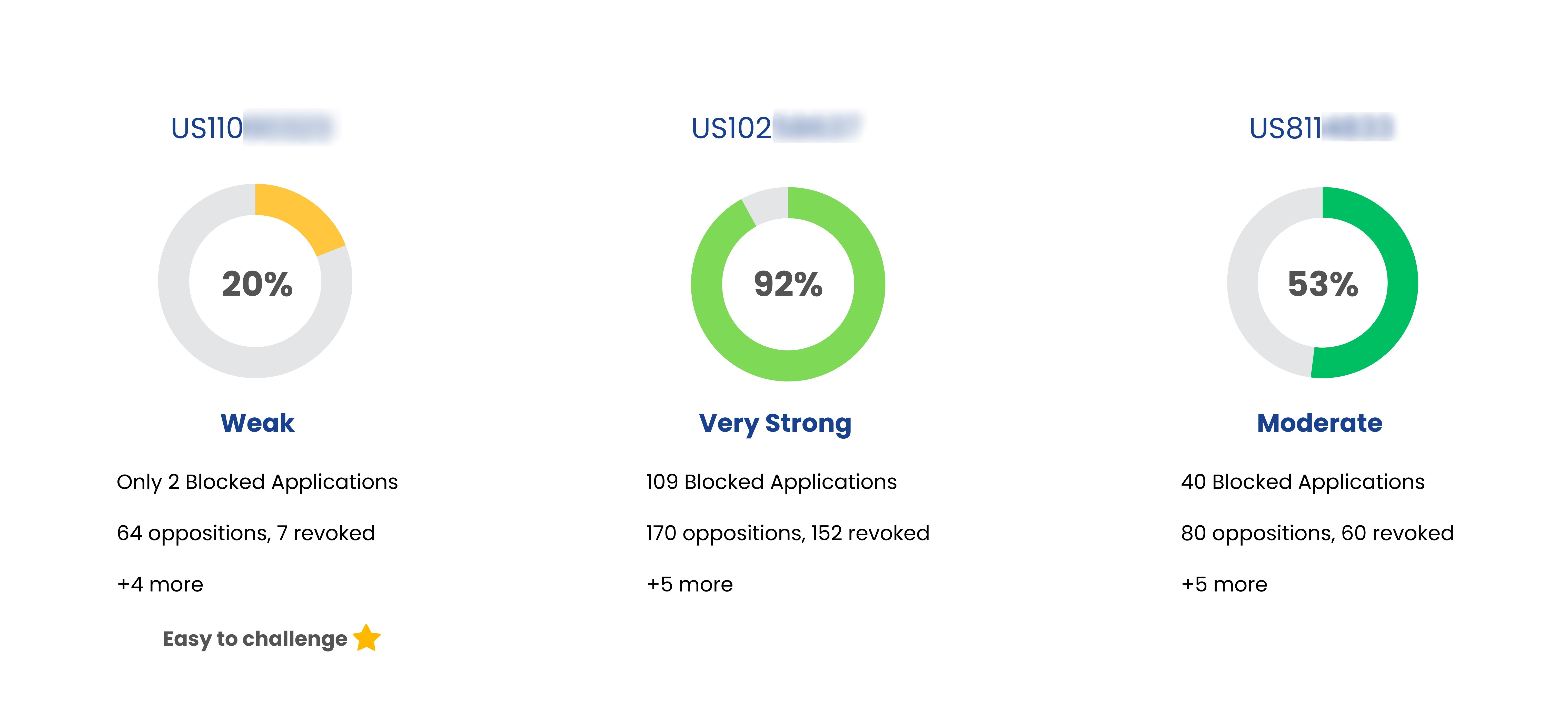
| Ciprodex | US6284804 | Topical suspension formulations containing ciprofloxacin and dexamethasone |
Aug 10, 2020
(Expired) | Sandoz |
| Ciprodex | US6359016 | Topical suspension formulations containing ciprofloxacin and dexamethasone |
Aug 10, 2020
(Expired) | Sandoz |
| Ciprodex | US8846650 | Method of treating middle ear infections | Jun 04, 2025 | Sandoz |
| Ciprodex | US9149486 | Method of treating middle ear infections |
Sep 13, 2022
(Expired) | Sandoz |
| Ciprodex | US9345714 | Method of treating middle ear infections |
Sep 13, 2022
(Expired) | Sandoz |
| Ciprodex | US9402805 | Method of treating middle ear infections |
Sep 13, 2022
(Expired) | Sandoz |
Coming Soon
Patent Strength Analyzer
YesNo
Thank you for your response 🥳
Ciprofloxacin; Dexamethasone Generics
Several generic applications have been filed for Ciprofloxacin; Dexamethasone. The first generic version for Ciprofloxacin; Dexamethasone was by Dr Reddys Laboratories Ltd and was approved on Aug 10, 2020. And the latest generic version is by Amneal Eu Ltd and was approved on Mar 22, 2024.
Given below is the list of companies who have filed for Ciprofloxacin; Dexamethasone generic.
1. SUN PHARM
Sun Pharmaceutical Industries Ltd has filed for 1 generic for Ciprofloxacin; Dexamethasone. This 0.3%;0.1% version comes by the name CIPROFLOXACIN AND DEXAMETHASONE. Given below are the details of the strengths of this generic introduced by Sun Pharm.
| Strength | Dosage Form | Availability | Application Pathway | TE code | Launch Date |
|---|---|---|---|---|---|
| 0.3%; 0.1% | suspension/drops | Prescription | OTIC | AB | Aug 30, 2022 |
2. DR REDDYS
Dr Reddys Laboratories Ltd has filed for 1 generic for Ciprofloxacin; Dexamethasone. This 0.3%;0.1% version comes by the name CIPROFLOXACIN AND DEXAMETHASONE. Given below are the details of the strengths of this generic introduced by Dr Reddys.
| Strength | Dosage Form | Availability | Application Pathway | TE code | Launch Date |
|---|---|---|---|---|---|
| 0.3%; 0.1% | suspension/drops | Prescription | OTIC | AB | Aug 10, 2020 |
3. SENTISS
Sentiss Ag has filed for 1 generic for Ciprofloxacin; Dexamethasone. This 0.3%;0.1% version comes by the name CIPROFLOXACIN AND DEXAMETHASONE. Given below are the details of the strengths of this generic introduced by Sentiss.
| Strength | Dosage Form | Availability | Application Pathway | TE code | Launch Date |
|---|---|---|---|---|---|
| 0.3%; 0.1% | suspension/drops | Prescription | OTIC | AB | Jun 9, 2023 |
4. AMNEAL
Amneal Eu Ltd has filed for 1 generic for Ciprofloxacin; Dexamethasone. Given below are the details of the strengths of this generic introduced by Amneal.
| Strength | Dosage Form | Availability | Application Pathway | TE code | Launch Date |
|---|---|---|---|---|---|
| 0.3%; 0.1% | suspension/drops | Prescription | OTIC | AB | Mar 22, 2024 |
