Insulin Lispro Recombinant Patent Expiration
Insulin Lispro Recombinant is Used for managing diabetes, irritable bowel syndrome with diarrhea, and hyperglycemia. It was first introduced by Eli Lilly And Co
Insulin Lispro Recombinant Patents
Given below is the list of patents protecting Insulin Lispro Recombinant, along with the drug name that holds that patent and the company name owning that drug.
| Drug Used in | Drug Patent Number | Drug Patent Title | Drug Patent Expiry | Drug Owner |
|---|---|---|---|---|
| Humalog | US5474978 | Insulin analog formulations |
Jun 16, 2014
(Expired) | Lilly |
| Humalog | US5514646 | Insulin analogs modified at position 29 of the B chain |
May 07, 2013
(Expired) | Lilly |
| Humalog Kwikpen | US5474978 | Insulin analog formulations |
Jun 16, 2014
(Expired) | Lilly |
| Humalog Kwikpen | US5514646 | Insulin analogs modified at position 29 of the B chain |
May 07, 2013
(Expired) | Lilly |
| Humalog Kwikpen | US6034054 | Stable insulin formulations |
Jun 11, 2018
(Expired) | Eli Lilly And Co |
| Humalog Kwikpen | US6551992 | Stable insulin formulations |
Jun 11, 2018
(Expired) | Eli Lilly And Co |
| Humalog Kwikpen | US7291132 | Medication dispensing apparatus with triple screw threads for mechanical advantage | Aug 09, 2024 | Eli Lilly And Co |
| Humalog Kwikpen | US7291132 | Medication dispensing apparatus with triple screw threads for mechanical advantage | Aug 09, 2024 | Lilly |
| Humalog Pen | US5474978 | Insulin analog formulations |
Jun 16, 2014
(Expired) | Lilly |
| Humalog Pen | US5514646 | Insulin analogs modified at position 29 of the B chain |
May 07, 2013
(Expired) | Lilly |
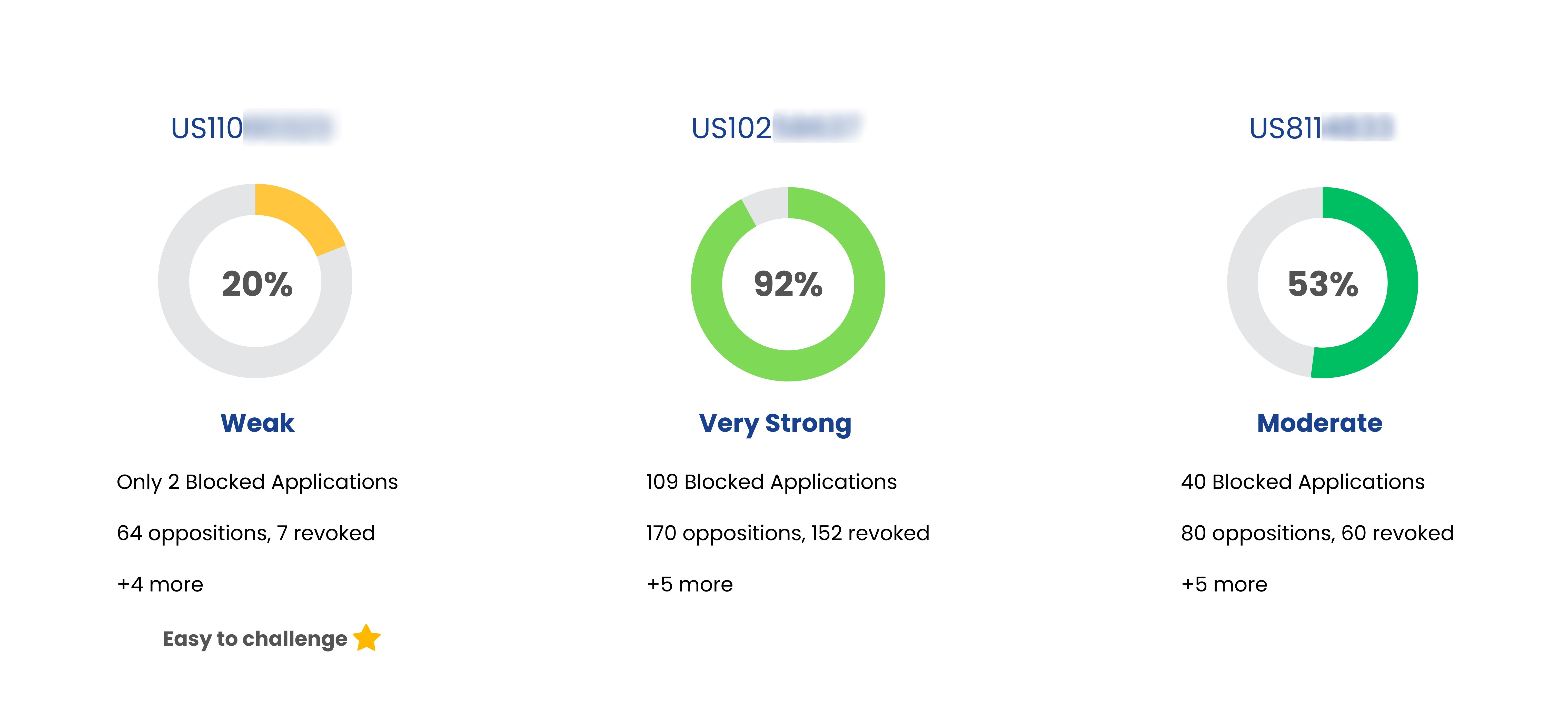
Coming Soon
Patent Strength Analyzer
YesNo
Thank you for your response 🥳
