Drospirenone; Estetrol Patent Expiration
Drospirenone; Estetrol is Used for contraception in females of reproductive potential. It was first introduced by Mayne Pharma Llc
Drospirenone; Estetrol Patents
Given below is the list of patents protecting Drospirenone; Estetrol, along with the drug name that holds that patent and the company name owning that drug.
| Drug Used in | Drug Patent Number | Drug Patent Title | Drug Patent Expiry | Drug Owner |
|---|---|---|---|---|
| Nextstellis | US11793760 | Orodispersible dosage unit containing an estetrol component | Jun 17, 2036 | Mayne Pharma |
| Nextstellis | US11957694 | Orodispersible dosage unit containing an estetrol component | Jun 17, 2036 | Mayne Pharma |
| Nextstellis | US11964055 | Orodispersible dosage unit containing an estetrol component | Jun 17, 2036 | Mayne Pharma |
| Nextstellis | US7732430 | Drug delivery system comprising a tetrahydroxilated estrogen for use in hormonal contraception | Mar 02, 2025 | Mayne Pharma |
Coming Soon
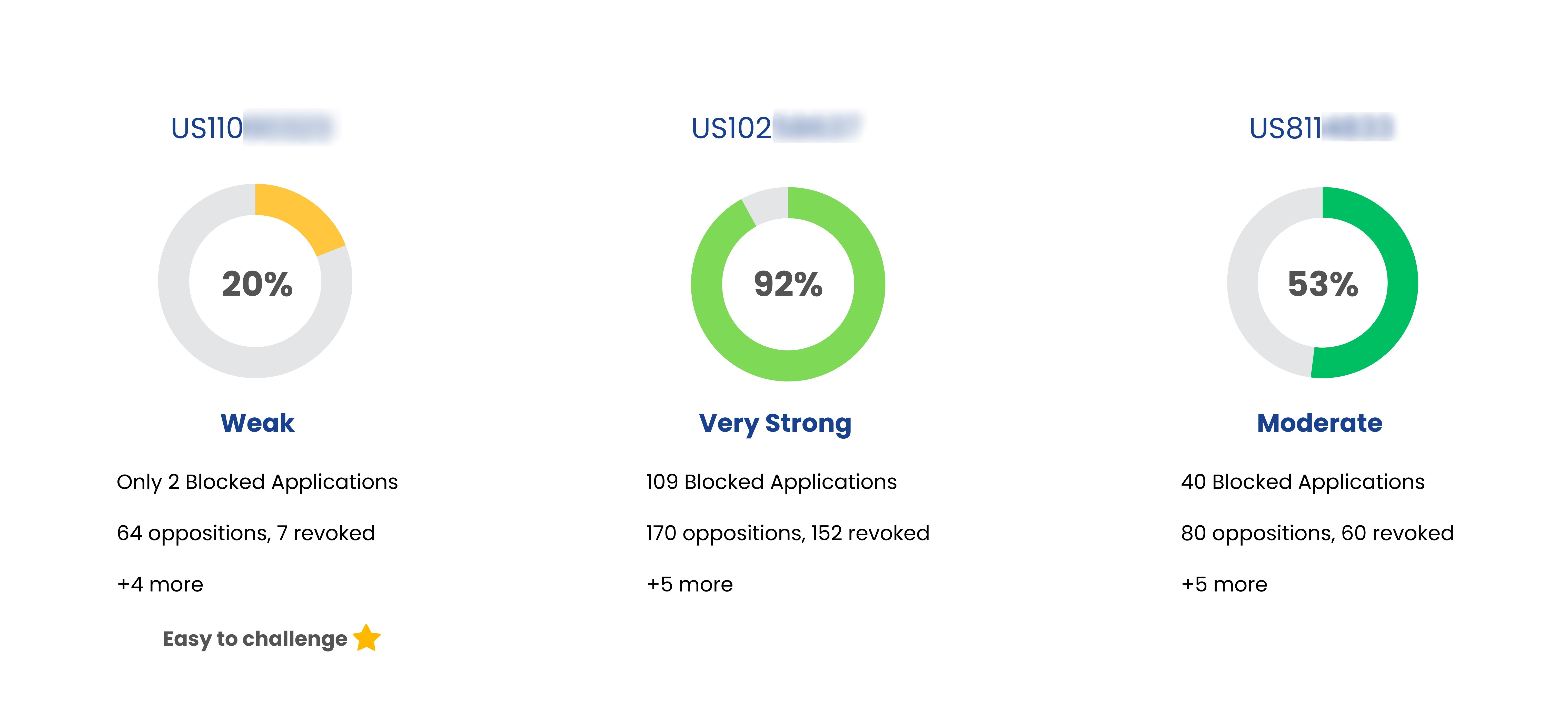
Patent Strength Analyzer
YesNo
Thank you for your response 🥳
