Budesonide; Formoterol Fumarate; Glycopyrrolate Patent Expiration
Budesonide; Formoterol Fumarate; Glycopyrrolate is Used for managing chronic obstructive pulmonary disease (COPD). It was first introduced by Astrazeneca Ab
Budesonide; Formoterol Fumarate; Glycopyrrolate Patents
Given below is the list of patents protecting Budesonide; Formoterol Fumarate; Glycopyrrolate, along with the drug name that holds that patent and the company name owning that drug.
| Drug Used in | Drug Patent Number | Drug Patent Title | Drug Patent Expiry | Drug Owner |
|---|---|---|---|---|
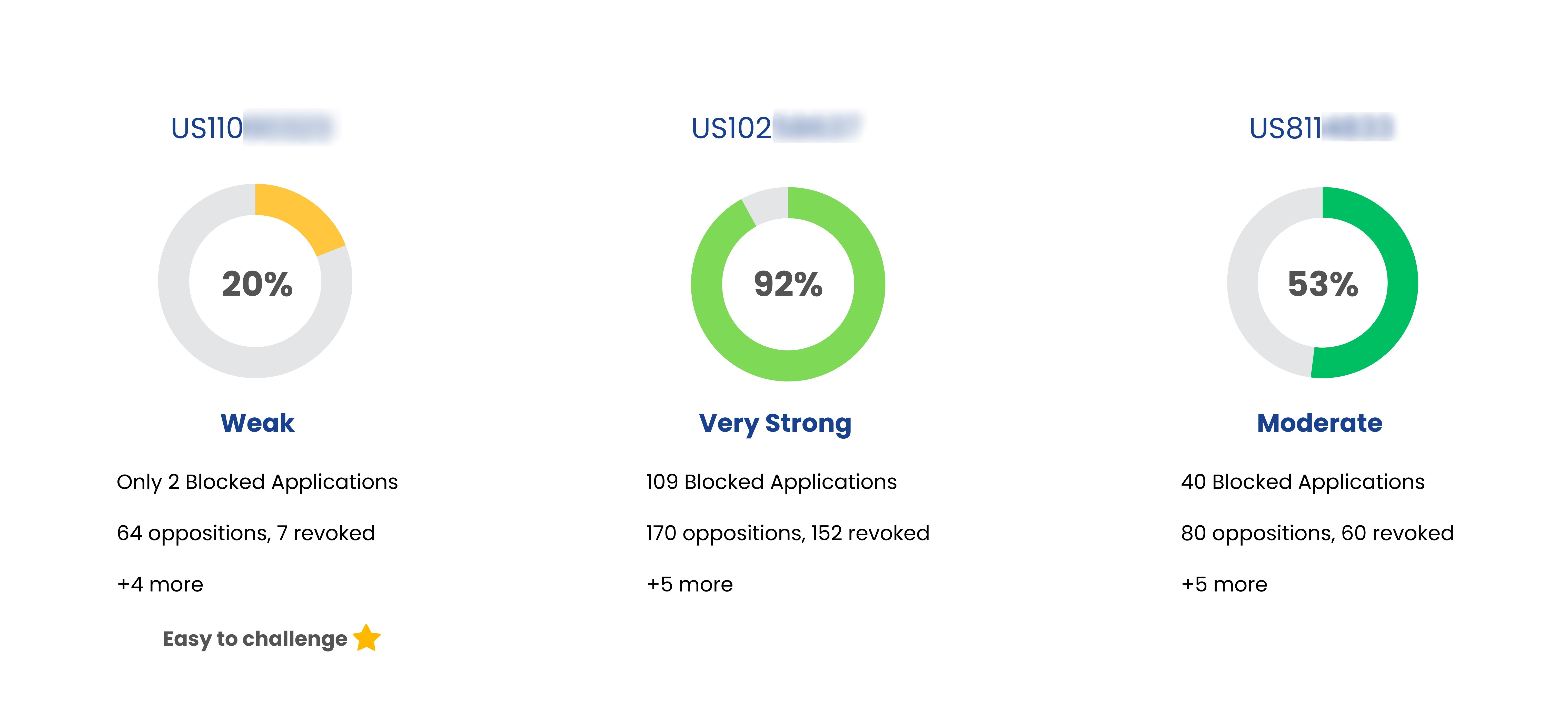
| Breztri Aerosphere | US10716753 | Compositions for pulmonary delivery of long-acting muscarinic antagonists or long-acting B2 adrenergic receptor agonists and associated methods and systems | May 28, 2030 | Astrazeneca Ab |
| Breztri Aerosphere | US11331442 | Drug delivery systems and related methods | Oct 05, 2038 | Astrazeneca Ab |
| Breztri Aerosphere | US11833292 | Drug delivery systems and related methods | Oct 05, 2038 | Astrazeneca Ab |
| Breztri Aerosphere | US8324266 | Compositions, methods and systems for respiratory delivery of two or more active agents | May 28, 2030 | Astrazeneca Ab |
| Breztri Aerosphere | US8703806 | Compositions, methods and propellant-based systems for respiratory delivery of glycopyrrolate and one or more active agents | May 28, 2030 | Astrazeneca Ab |
| Breztri Aerosphere | US8808713 | Compositions for pulmonary delivery of long-acting β2 adrenergic receptor agonists and associated methods and systems | May 28, 2030 | Astrazeneca Ab |
| Breztri Aerosphere | US8815258 | Compositions, methods and systems for respiratory delivery of two or more active agents | Mar 17, 2031 | Astrazeneca Ab |
| Breztri Aerosphere | US9415009 | Compositions, methods and systems for respiratory delivery of two or more active agents | May 28, 2030 | Astrazeneca Ab |
| Breztri Aerosphere | US9463161 | Compositions for pulmonary delivery of long-acting muscarinic antagonists and associated methods and systems | May 28, 2030 | Astrazeneca Ab |
Coming Soon
Patent Strength Analyzer
YesNo
Thank you for your response 🥳
