Sacubitril; Valsartan Patent Expiration
Sacubitril; Valsartan is Used for treating heart failure with reduced ejection fraction in patients not taking ACE inhibitors or ARBs, or previously on low doses, by titrating up from half the usual starting dose. It was first introduced by Novartis Pharmaceuticals Corp
Sacubitril; Valsartan Patents
Given below is the list of patents protecting Sacubitril; Valsartan, along with the drug name that holds that patent and the company name owning that drug.
| Drug Used in | Drug Patent Number | Drug Patent Title | Drug Patent Expiry | Drug Owner |
|---|---|---|---|---|
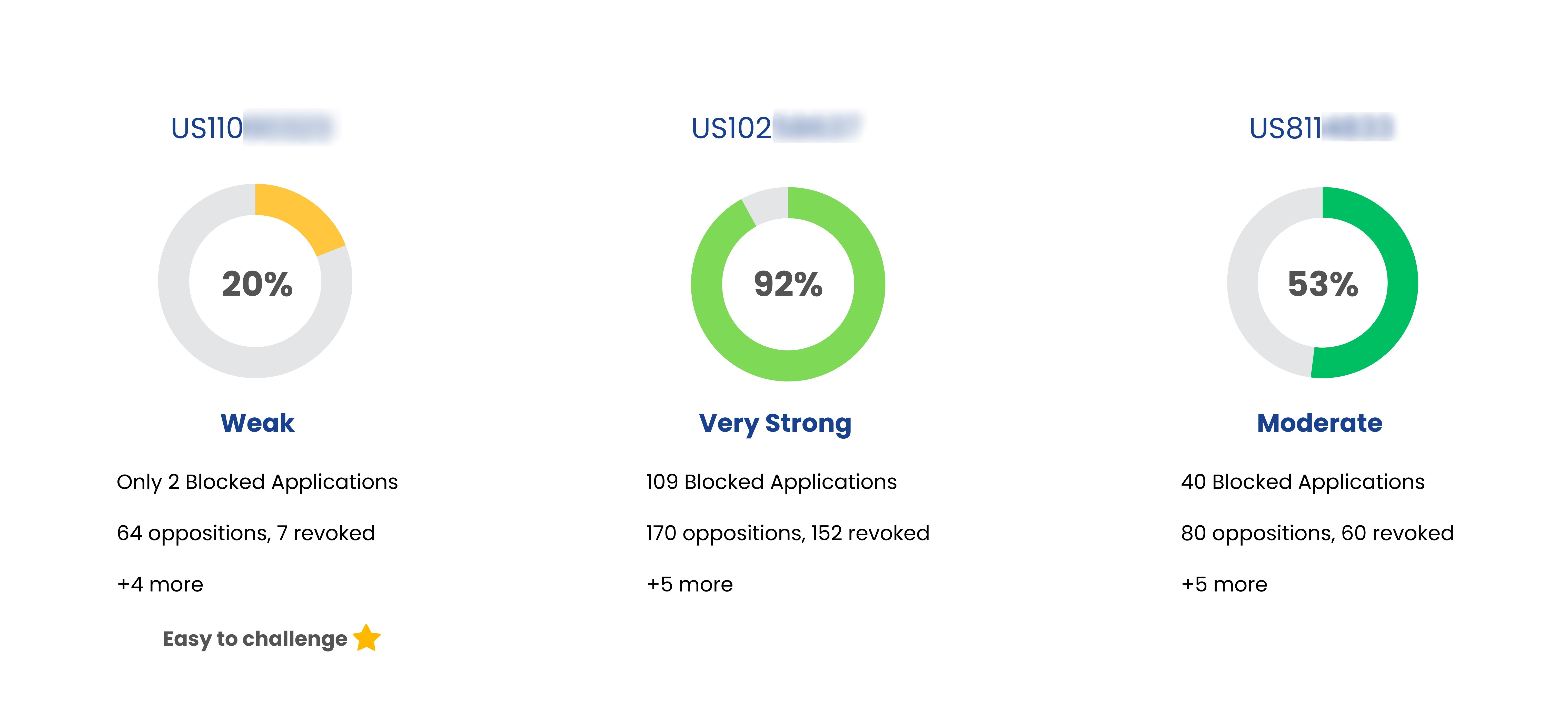
| Entresto | US11058667 | Sacubitril-valsartan dosage regimen for treating heart failure | May 09, 2036 | Novartis Pharms Corp |
| Entresto | US11135192 | Inhibitors for treating diseases characterized by atrial enlargement or remodeling | Aug 22, 2033 | Novartis Pharms Corp |
| Entresto | US7468390 | Methods of treatment and pharmaceutical composition |
Nov 27, 2023
(Expired) | Novartis Pharms Corp |
| Entresto |
US7468390 (Pediatric) | Methods of treatment and pharmaceutical composition |
May 27, 2024
(Expired) | Novartis Pharms Corp |
| Entresto | US8101659 | Methods of treatment and pharmaceutical composition | Jan 15, 2025 | Novartis Pharms Corp |
| Entresto |
US8101659 (Pediatric) | Methods of treatment and pharmaceutical composition | Jul 15, 2025 | Novartis Pharms Corp |
| Entresto | US8404744 | Methods of treatment and pharmaceutical composition |
Jan 14, 2023
(Expired) | Novartis Pharms Corp |
| Entresto |
US8404744 (Pediatric) | Methods of treatment and pharmaceutical composition |
Jul 14, 2023
(Expired) | Novartis Pharms Corp |
| Entresto | US8796331 | Methods of treatment and pharmaceutical composition |
Jan 14, 2023
(Expired) | Novartis Pharms Corp |
| Entresto |
US8796331 (Pediatric) | Methods of treatment and pharmaceutical composition |
Jul 14, 2023
(Expired) | Novartis Pharms Corp |
| Entresto | US8877938 | Compounds containing S-N-valeryl-N-{[2′-(1H-tetrazole-5-yl)-biphenyl-4-yl]-methyl}-valine and (2R,4S)-5-biphenyl-4-yl-4-(3-carboxy-propionylamino)-2-methyl-pentanoic acid ethyl ester moieties and cations | May 27, 2027 | Novartis Pharms Corp |
| Entresto |
US8877938 (Pediatric) | Compounds containing S-N-valeryl-N-{[2′-(1H-tetrazole-5-yl)-biphenyl-4-yl]-methyl}-valine and (2R,4S)-5-biphenyl-4-yl-4-(3-carboxy-propionylamino)-2-methyl-pentanoic acid ethyl ester moieties and cations | Nov 27, 2027 | Novartis Pharms Corp |
| Entresto | US9388134 | Compounds containing S-N-valeryl-N-{[2′-(1H-tetrazole-5-yl)-biphenyl-4-yl]-methyl)-valine and (2R,4S)-5-biphenyl-4-yl-4-(3-carboxy-propionylamino)-2-methyl-pentanoic acid ethyl ester moieties and cations | Nov 08, 2026 | Novartis Pharms Corp |
| Entresto |
US9388134 (Pediatric) | Compounds containing S-N-valeryl-N-{[2′-(1H-tetrazole-5-yl)-biphenyl-4-yl]-methyl)-valine and (2R,4S)-5-biphenyl-4-yl-4-(3-carboxy-propionylamino)-2-methyl-pentanoic acid ethyl ester moieties and cations | May 08, 2027 | Novartis Pharms Corp |
| Entresto | US9517226 | Inhibitors for treating diseases characterized by atrial enlargement or remodeling | Aug 22, 2033 | Novartis Pharms Corp |
| Entresto | US9937143 | Inhibitors for treating diseases characterized by atrial enlargement or remodeling | Aug 22, 2033 | Novartis Pharms Corp |
| Entresto Sprinkle | US10722471 | Galenic formulations of organic compounds | Feb 02, 2037 | Novartis |
| Entresto Sprinkle | US8101659 | Methods of treatment and pharmaceutical composition | Jan 15, 2025 | Novartis |
| Entresto Sprinkle | US8877938 | Compounds containing S-N-valeryl-N-{[2′-(1H-tetrazole-5-yl)-biphenyl-4-yl]-methyl}-valine and (2R,4S)-5-biphenyl-4-yl-4-(3-carboxy-propionylamino)-2-methyl-pentanoic acid ethyl ester moieties and cations | May 27, 2027 | Novartis |
| Entresto Sprinkle | US9388134 | Compounds containing S-N-valeryl-N-{[2′-(1H-tetrazole-5-yl)-biphenyl-4-yl]-methyl)-valine and (2R,4S)-5-biphenyl-4-yl-4-(3-carboxy-propionylamino)-2-methyl-pentanoic acid ethyl ester moieties and cations | Nov 08, 2026 | Novartis |
Coming Soon
Patent Strength Analyzer
YesNo
Thank you for your response 🥳
Sacubitril; Valsartan Generics
Several generic applications have been filed for Sacubitril; Valsartan. The first generic version for Sacubitril; Valsartan was by Crystal Pharmaceutical Suzhou Co Ltd and was approved on May 28, 2024. And the latest generic version is by Zydus Pharmaceuticals Usa Inc and was approved on Jul 9, 2024.
Given below is the list of companies who have filed for Sacubitril; Valsartan generic.
1. CRYSTAL
Crystal Pharmaceutical Suzhou Co Ltd has filed for 3 different strengths of generic version for Sacubitril; Valsartan. All of these versions come by the name SACUBITRIL AND VALSARTAN. Given below are the details of the strengths of this generic introduced by Crystal.
| Strength | Dosage Form | Availability | Application Pathway | TE code | Launch Date |
|---|---|---|---|---|---|
| 49MG; 51MG | tablet | Prescription | ORAL | AB | May 28, 2024 |
| 24MG; 26MG | tablet | Prescription | ORAL | AB | May 28, 2024 |
| 97MG; 103MG | tablet | Prescription | ORAL | AB | May 28, 2024 |
2. LAURUS
Laurus Labs Ltd has filed for 3 different strengths of generic version for Sacubitril; Valsartan. All of these versions come by the name SACUBITRIL AND VALSARTAN. Given below are the details of the strengths of this generic introduced by Laurus.
| Strength | Dosage Form | Availability | Application Pathway | TE code | Launch Date |
|---|---|---|---|---|---|
| 24MG; 26MG | tablet | Prescription | ORAL | AB | May 28, 2024 |
| 49MG; 51MG | tablet | Prescription | ORAL | AB | May 28, 2024 |
| 97MG; 103MG | tablet | Prescription | ORAL | AB | May 28, 2024 |
3. ZYDUS PHARMS
Zydus Pharmaceuticals Usa Inc has filed for 3 different strengths of generic version for Sacubitril; Valsartan. All of these versions come by the name SACUBITRIL AND VALSARTAN. Given below are the details of the strengths of this generic introduced by Zydus Pharms.
| Strength | Dosage Form | Availability | Application Pathway | TE code | Launch Date |
|---|---|---|---|---|---|
| 97MG; 103MG | tablet | Prescription | ORAL | AB | Jul 9, 2024 |
| 24MG; 26MG | tablet | Prescription | ORAL | AB | Jul 9, 2024 |
| 49MG; 51MG | tablet | Prescription | ORAL | AB | Jul 9, 2024 |
4. ALEMBIC
Alembic Pharmaceuticals Ltd has filed for 3 different strengths of generic version for Sacubitril; Valsartan. All of these versions come by the name SACUBITRIL AND VALSARTAN. Given below are the details of the strengths of this generic introduced by Alembic.
| Strength | Dosage Form | Availability | Application Pathway | TE code | Launch Date |
|---|---|---|---|---|---|
| 24MG; 26MG | tablet | Prescription | ORAL | AB | May 28, 2024 |
| 49MG; 51MG | tablet | Prescription | ORAL | AB | May 28, 2024 |
| 97MG; 103MG | tablet | Prescription | ORAL | AB | May 28, 2024 |
