Ombitasvir; Paritaprevir; Ritonavir Patent Expiration
Ombitasvir; Paritaprevir; Ritonavir is Used for treating Hepatitis C virus (HCV) infection. It was first introduced by Abbvie Inc
Ombitasvir; Paritaprevir; Ritonavir Patents
Given below is the list of patents protecting Ombitasvir; Paritaprevir; Ritonavir, along with the drug name that holds that patent and the company name owning that drug.
| Drug Used in | Drug Patent Number | Drug Patent Title | Drug Patent Expiry | Drug Owner |
|---|---|---|---|---|
| Technivie | US6037157 | Method for improving pharmacokinetics |
Jun 26, 2016
(Expired) | Abbvie |
| Technivie |
US6037157 (Pediatric) | Method for improving pharmacokinetics |
Dec 26, 2016
(Expired) | Abbvie |
| Technivie | US6703403 | Method for improving pharmacokinetics |
Jun 26, 2016
(Expired) | Abbvie |
| Technivie |
US6703403 (Pediatric) | Method for improving pharmacokinetics |
Dec 26, 2016
(Expired) | Abbvie |
| Technivie | US7148359 | Polymorph of a pharmaceutical |
Jul 19, 2019
(Expired) | Abbvie |
| Technivie |
US7148359 (Pediatric) | Polymorph of a pharmaceutical |
Jan 19, 2020
(Expired) | Abbvie |
| Technivie | US7364752 | Solid dispersion pharamaceutical formulations |
Nov 10, 2020
(Expired) | Abbvie |
| Technivie |
US7364752 (Pediatric) | Solid dispersion pharamaceutical formulations |
May 10, 2021
(Expired) | Abbvie |
| Technivie | US8268349 | Solid pharmaceutical dosage form | Aug 25, 2024 | Abbvie |
| Technivie |
US8268349 (Pediatric) | Solid pharmaceutical dosage form | Feb 25, 2025 | Abbvie |
| Technivie | US8399015 | Solid pharmaceutical dosage form | Aug 25, 2024 | Abbvie |
| Technivie |
US8399015 (Pediatric) | Solid pharmaceutical dosage form | Feb 25, 2025 | Abbvie |
| Technivie | US8420596 | Macrocyclic hepatitis C serine protease inhibitors | Apr 10, 2031 | Abbvie |
| Technivie |
US8420596 (Pediatric) | Macrocyclic hepatitis C serine protease inhibitors | Oct 10, 2031 | Abbvie |
| Technivie | US8642538 | Macrocyclic hepatitis C serine protease inhibitors | Sep 10, 2029 | Abbvie |
| Technivie | US8686026 | Solid compositions | Jun 09, 2031 | Abbvie |
| Technivie | US8691938 | Anti-viral compounds | Apr 13, 2032 | Abbvie |
| Technivie | US9006387 | Anti-viral compounds | Jun 10, 2030 | Abbvie |
| Technivie | US9044480 | Compositions and methods for treating HCV | Apr 10, 2031 | Abbvie |
A patent's expiry date may change depending upon legal activities going on that patent. Critical activities like abandoning of a patent, term extension of a patent or amendment of its claims can increase or decrease the life of a patent hence affecting its expiry date and in turn affecting the generic launch date of that drug. Tracking these ongoing activities on a patent application helps to keep an eye on the latest developments in the patent process of the drug which can give an idea of how early a drug's generic could be available. The next section provides a list of recent legal activities on Ombitasvir; Paritaprevir; Ritonavir's patents.
Latest Legal Activities on Ombitasvir; Paritaprevir; Ritonavir's Patents
Given below is the list recent legal activities going on the following patents of Ombitasvir; Paritaprevir; Ritonavir.
| Event | Date | Patent/Publication |
|---|---|---|
 | ||
| Payment of Maintenance Fee, 8th Year, Large Entity | 09 Nov, 2022 | US9044480 |
| Payment of Maintenance Fee, 8th Year, Large Entity | 14 Sep, 2022 | US9006387 |
| Payment of Maintenance Fee, 8th Year, Large Entity | 09 Sep, 2021 | US8691938 |
| Payment of Maintenance Fee, 8th Year, Large Entity | 09 Sep, 2021 | US8686026 |
| Payment of Maintenance Fee, 8th Year, Large Entity | 15 Jul, 2021 | US8642538 |
| Payment of Maintenance Fee, 8th Year, Large Entity | 15 Sep, 2020 | US8420596 |
| Payment of Maintenance Fee, 4th Year, Large Entity | 15 Nov, 2018 | US9044480 |
| Payment of Maintenance Fee, 4th Year, Large Entity | 13 Sep, 2018 | US9006387 |
| Payment of Maintenance Fee, 4th Year, Large Entity | 14 Sep, 2017 | US8686026 |
| Payment of Maintenance Fee, 4th Year, Large Entity | 14 Sep, 2017 | US8691938 |
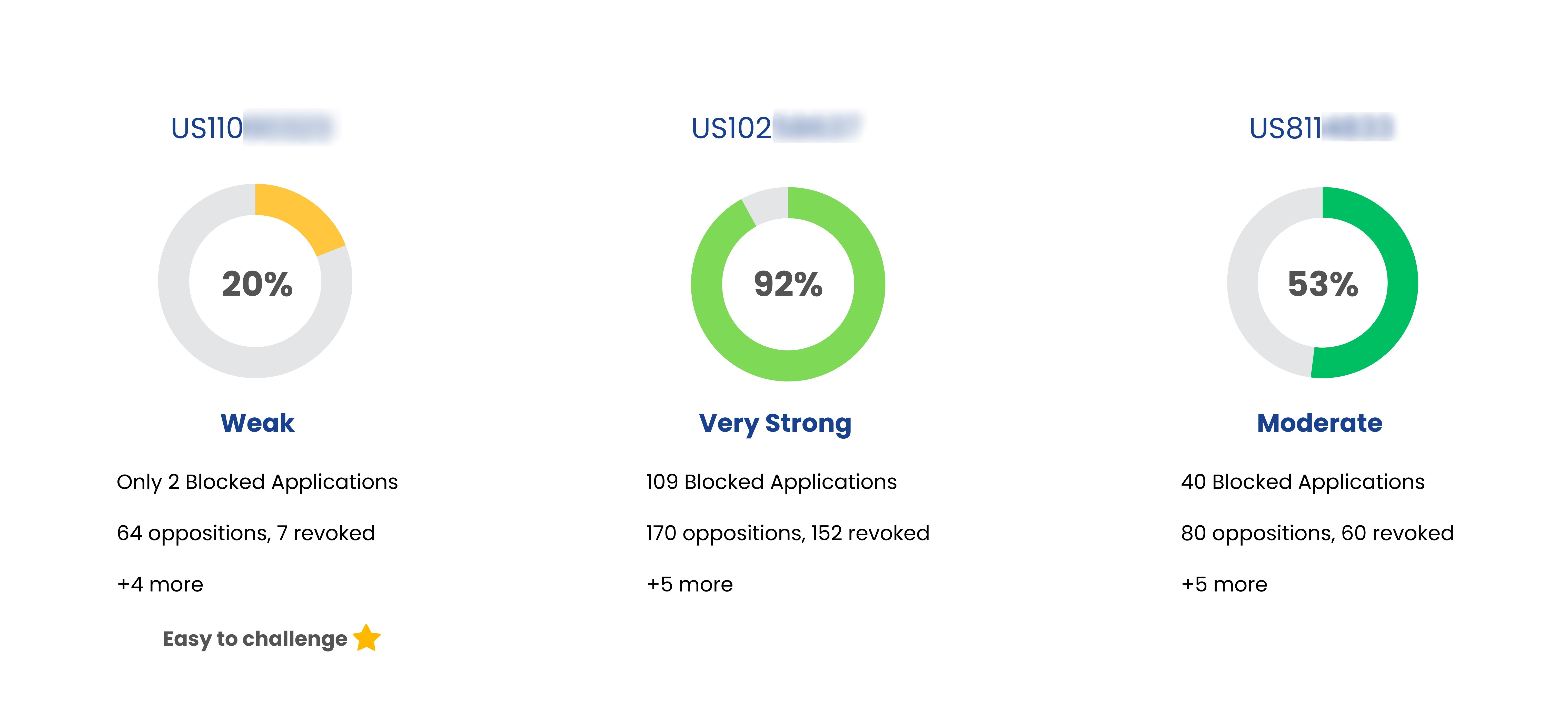
Coming Soon
Patent Strength Analyzer
YesNo
Thank you for your response 🥳
