Mecasermin Recombinant Patent Expiration
Mecasermin Recombinant is Used for treating primary IGF-1 deficiency. It was first introduced by Ipsen Biopharmaceuticals Inc
Mecasermin Recombinant Patents
Given below is the list of patents protecting Mecasermin Recombinant, along with the drug name that holds that patent and the company name owning that drug.
| Drug Used in | Drug Patent Number | Drug Patent Title | Drug Patent Expiry | Drug Owner |
|---|---|---|---|---|
| Increlex | US5681814 | Formulated IGF-I Composition |
Sep 18, 2017
(Expired) | Ipsen Inc |
| Increlex | US5824642 | Treatment of partial growth hormone insensitivity syndrome |
Jul 08, 2014
(Expired) | Ipsen Inc |
| Increlex | US6207640 | Treatment of partial growth hormone insensitivity syndrome |
Apr 07, 2014
(Expired) | Ipsen Inc |
Coming Soon
Patent Strength Analyzer
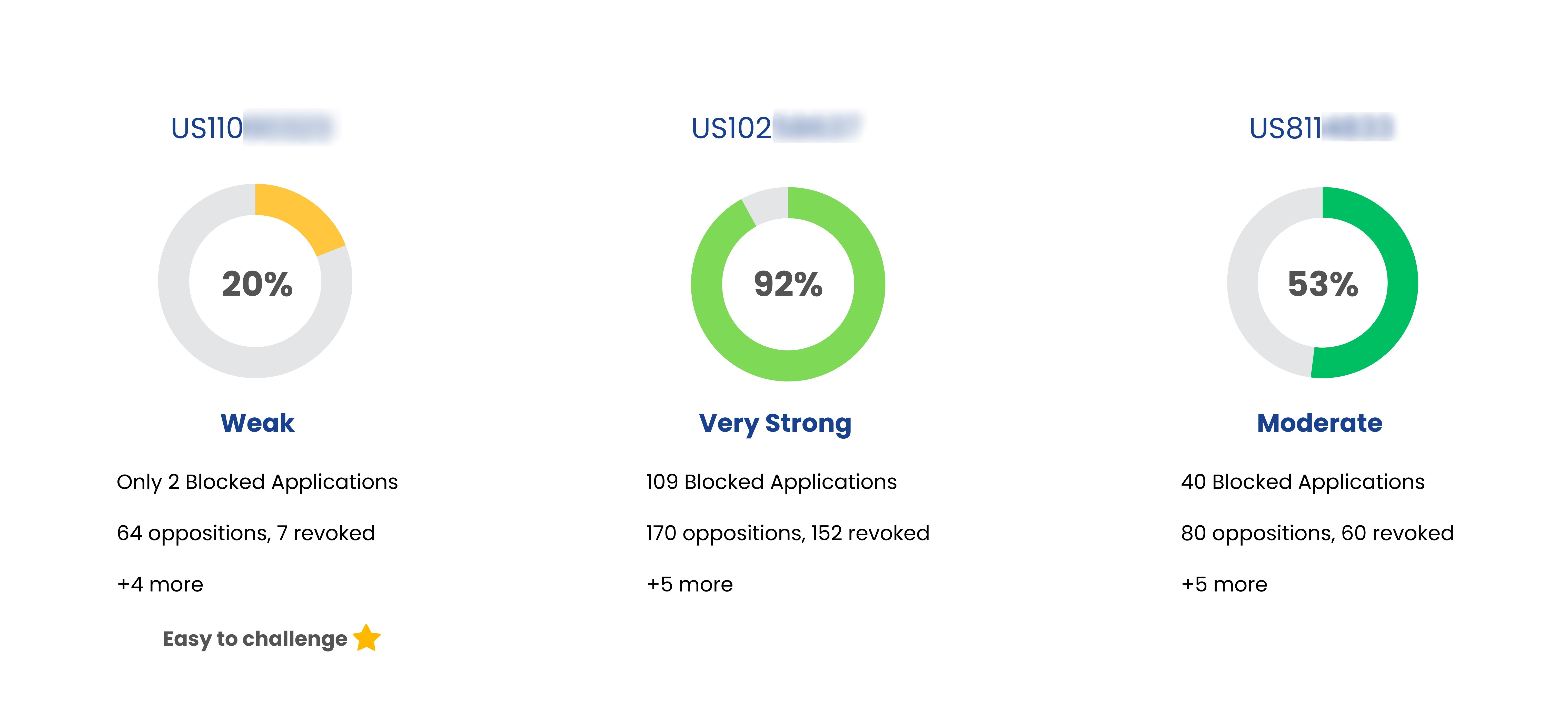
YesNo
Thank you for your response 🥳
