Ethinyl Estradiol; Segesterone Acetate Patent Expiration
Ethinyl Estradiol; Segesterone Acetate is used for hormonal contraception. It was first introduced by Mayne Pharma Llc
Ethinyl Estradiol; Segesterone Acetate Patents
Given below is the list of patents protecting Ethinyl Estradiol; Segesterone Acetate, along with the drug name that holds that patent and the company name owning that drug.
| Drug Used in | Drug Patent Number | Drug Patent Title | Drug Patent Expiry | Drug Owner |
|---|---|---|---|---|
| Annovera | US10632066 | Method of providing birth control | Feb 01, 2039 | Mayne Pharma |
| Annovera | US10765628 | Method of providing birth control | Feb 01, 2039 | Mayne Pharma |
| Annovera | US10780047 | Method of providing birth control | Feb 01, 2039 | Mayne Pharma |
| Annovera | US10918649 | System for providing birth control | Jun 21, 2039 | Mayne Pharma |
| Annovera | US10925882 | System for providing birth control | Jun 21, 2039 | Mayne Pharma |
| Annovera | US10940157 | System for providing birth control | Jun 21, 2039 | Mayne Pharma |
| Annovera | US11529308 | System for providing birth control | Jun 21, 2039 | Mayne Pharma |
| Annovera | US11850251 | System for providing birth control | Jun 21, 2039 | Mayne Pharma |
Coming Soon
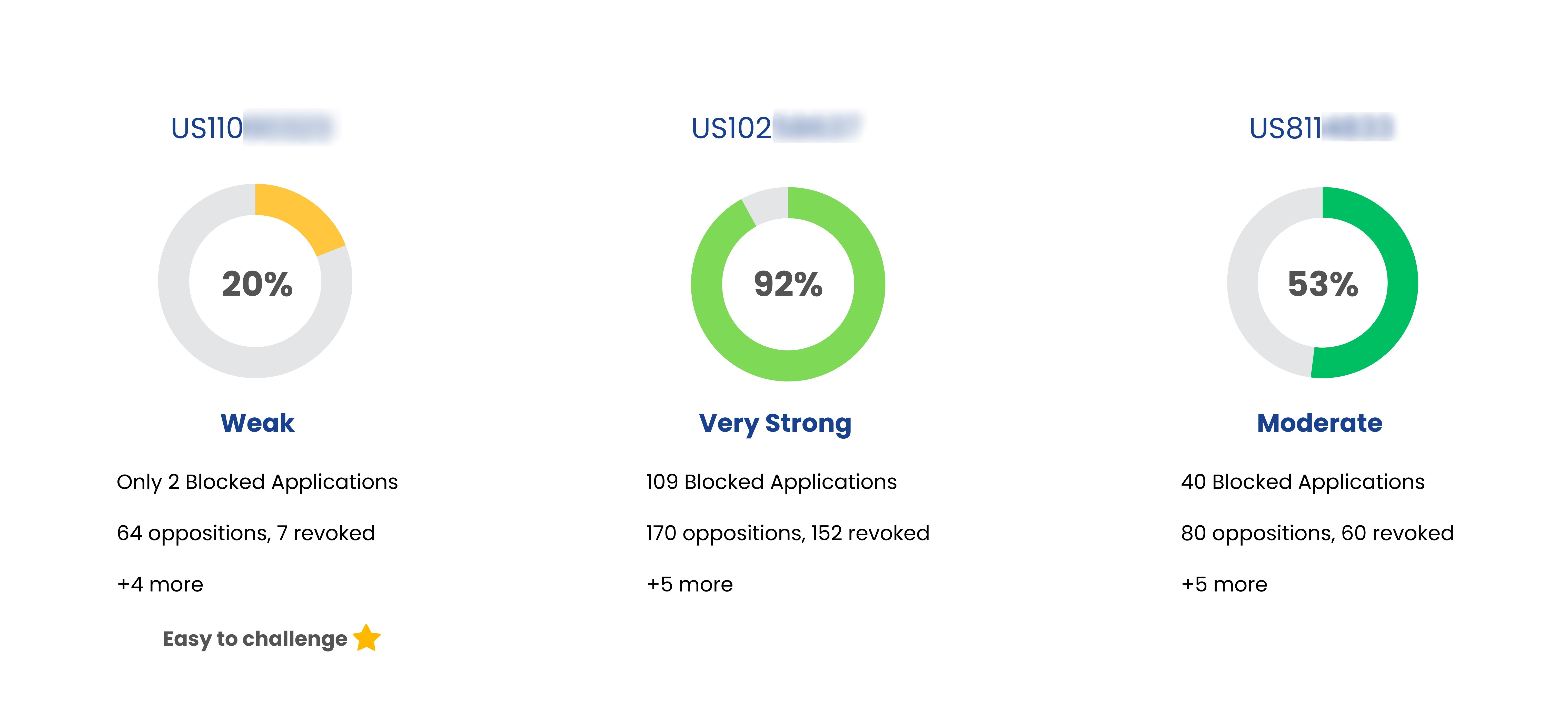
Patent Strength Analyzer
YesNo
Thank you for your response 🥳
