Doravirine; Lamivudine; Tenofovir Disoproxil Fumarate Patent Expiration
Doravirine; Lamivudine; Tenofovir Disoproxil Fumarate is used for treating HIV-1 infection in pediatric patients weighing at least 35kg without prior antiretroviral treatment or replacing current regimen in virologically suppressed patients. It was first introduced by Merck Sharp And Dohme Corp A Sub Of Merck And Co Inc
Doravirine; Lamivudine; Tenofovir Disoproxil Fumarate Patents
Given below is the list of patents protecting Doravirine; Lamivudine; Tenofovir Disoproxil Fumarate, along with the drug name that holds that patent and the company name owning that drug.
| Drug Used in | Drug Patent Number | Drug Patent Title | Drug Patent Expiry | Drug Owner |
|---|---|---|---|---|
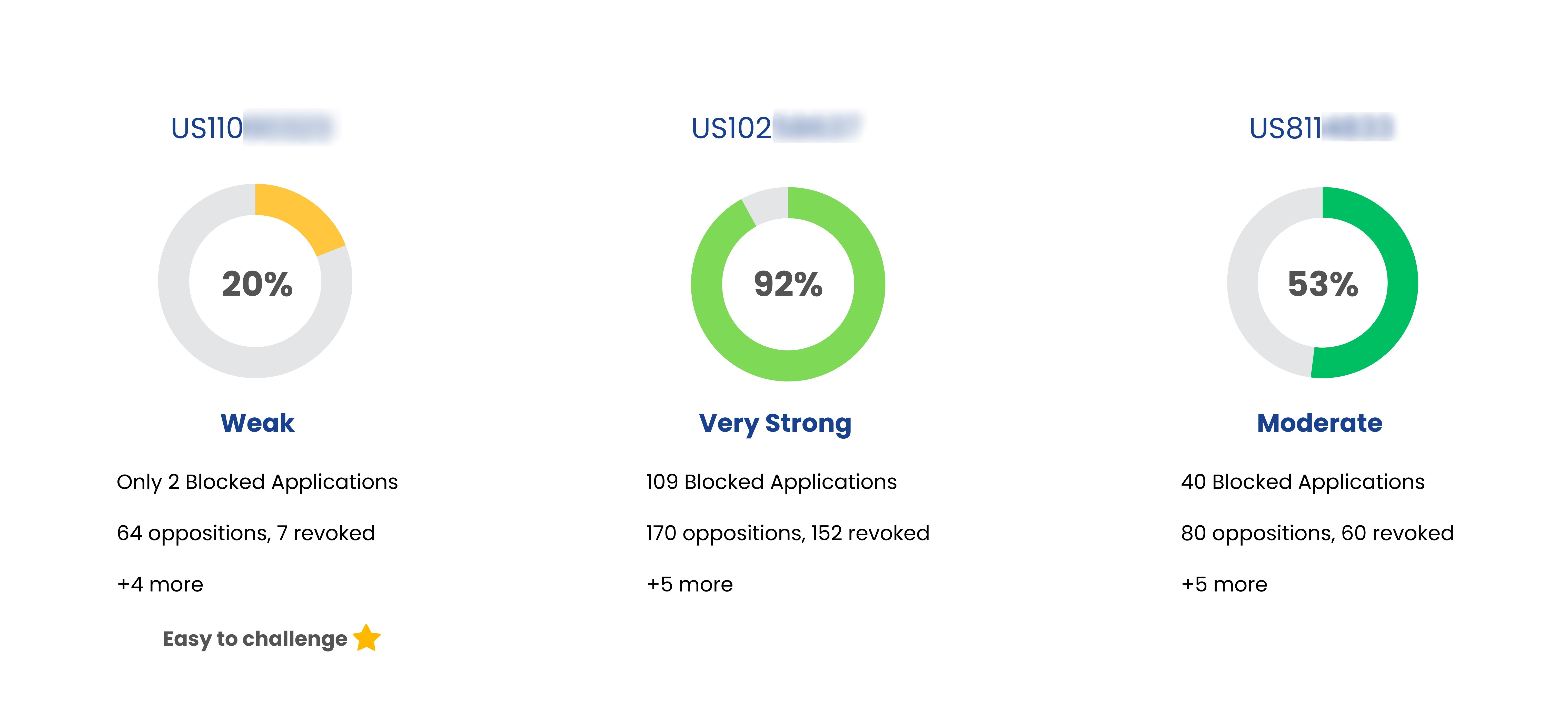
| Delstrigo | US10603282 | Pharmaceutical compositions containing doravirine, tenofovir disoproxil fumarate and lamivudine | Nov 29, 2036 | Msd Merck Co |
| Delstrigo | US10842751 | Pharmaceutical compositions containing doravirine, tenofovir disoproxil fumarate and lamivudine | Nov 29, 2036 | Msd Merck Co |
| Delstrigo | US8486975 | Non-nucleoside reverse transcriptase inhibitors | Oct 07, 2031 | Msd Merck Co |
Coming Soon
Patent Strength Analyzer
YesNo
Thank you for your response 🥳
