Dapagliflozin; Metformin Hydrochloride Patent Expiration
Dapagliflozin; Metformin Hydrochloride is used for managing type 2 diabetes mellitus when glycemic control is not achievable with other medications. It was first introduced by Astrazeneca Ab
Dapagliflozin; Metformin Hydrochloride Patents
Given below is the list of patents protecting Dapagliflozin; Metformin Hydrochloride, along with the drug name that holds that patent and the company name owning that drug.
| Drug Used in | Drug Patent Number | Drug Patent Title | Drug Patent Expiry | Drug Owner |
|---|---|---|---|---|
| Xigduo Xr | US6414126 | C-aryl glucoside SGLT2 inhibitors and method |
Oct 04, 2020
(Expired) | Astrazeneca Ab |
| Xigduo Xr | US6515117 | C-aryl glucoside SGLT2 inhibitors and method | Oct 04, 2025 | Astrazeneca Ab |
| Xigduo Xr |
US6515117 (Pediatric) | C-aryl glucoside SGLT2 inhibitors and method | Apr 04, 2026 | Astrazeneca Ab |
| Xigduo Xr | US6936590 | C-aryl glucoside SGLT2 inhibitors and method |
Oct 04, 2020
(Expired) | Astrazeneca Ab |
| Xigduo Xr | US7919598 | Crystal structures of SGLT2 inhibitors and processes for preparing same | Dec 16, 2029 | Astrazeneca Ab |
| Xigduo Xr |
US7919598 (Pediatric) | Crystal structures of SGLT2 inhibitors and processes for preparing same | Jun 16, 2030 | Astrazeneca Ab |
| Xigduo Xr | US8501698 | Crystal structures of SGLT2 inhibitors and processes for preparing same | Jun 20, 2027 | Astrazeneca Ab |
| Xigduo Xr |
US8501698 (Pediatric) | Crystal structures of SGLT2 inhibitors and processes for preparing same | Dec 20, 2027 | Astrazeneca Ab |
| Xigduo Xr | US8685934 | Methods for treating extreme insulin resistance in patients resistant to previous treatment with other anti-diabetic drugs employing an SGLT2 inhibitor and compositions thereof | May 26, 2030 | Astrazeneca Ab |
| Xigduo Xr |
US8685934 (Pediatric) | Methods for treating extreme insulin resistance in patients resistant to previous treatment with other anti-diabetic drugs employing an SGLT2 inhibitor and compositions thereof | Nov 26, 2030 | Astrazeneca Ab |
| Xigduo Xr | US9198925 | Pharmaceutical formulations containing an SGLT2 inhibitor |
Oct 04, 2020
(Expired) | Astrazeneca Ab |
| Xigduo Xr | US9616028 | Bilayer tablet formulations | Nov 12, 2030 | Astrazeneca Ab |
| Xigduo Xr |
US9616028 (Pediatric) | Bilayer tablet formulations | May 12, 2031 | Astrazeneca Ab |
A patent's expiry date may change depending upon legal activities going on that patent. Critical activities like abandoning of a patent, term extension of a patent or amendment of its claims can increase or decrease the life of a patent hence affecting its expiry date and in turn affecting the generic launch date of that drug. Tracking these ongoing activities on a patent application helps to keep an eye on the latest developments in the patent process of the drug which can give an idea of how early a drug's generic could be available. The next section provides a list of recent legal activities on Dapagliflozin; Metformin Hydrochloride's patents.
Latest Legal Activities on Dapagliflozin; Metformin Hydrochloride's Patents
Given below is the list recent legal activities going on the following patents of Dapagliflozin; Metformin Hydrochloride.
| Event | Date | Patent/Publication |
|---|---|---|
 | ||
| Payment of Maintenance Fee, 4th Year, Large Entity | 24 Sep, 2020 | US9616028 |
| Recordation of Patent Grant Mailed | 11 Apr, 2017 | US9616028 |
| Patent Issue Date Used in PTA Calculation | 11 Apr, 2017 | US9616028 |
| Email Notification | 23 Mar, 2017 | US9616028 |
| Issue Notification Mailed | 22 Mar, 2017 | US9616028 |
| Dispatch to FDC | 02 Mar, 2017 | US9616028 |
| Application Is Considered Ready for Issue | 02 Mar, 2017 | US9616028 |
| Issue Fee Payment Verified | 24 Feb, 2017 | US9616028 |
| Issue Fee Payment Received | 24 Feb, 2017 | US9616028 |
| Electronic Review | 28 Nov, 2016 | US9616028 |
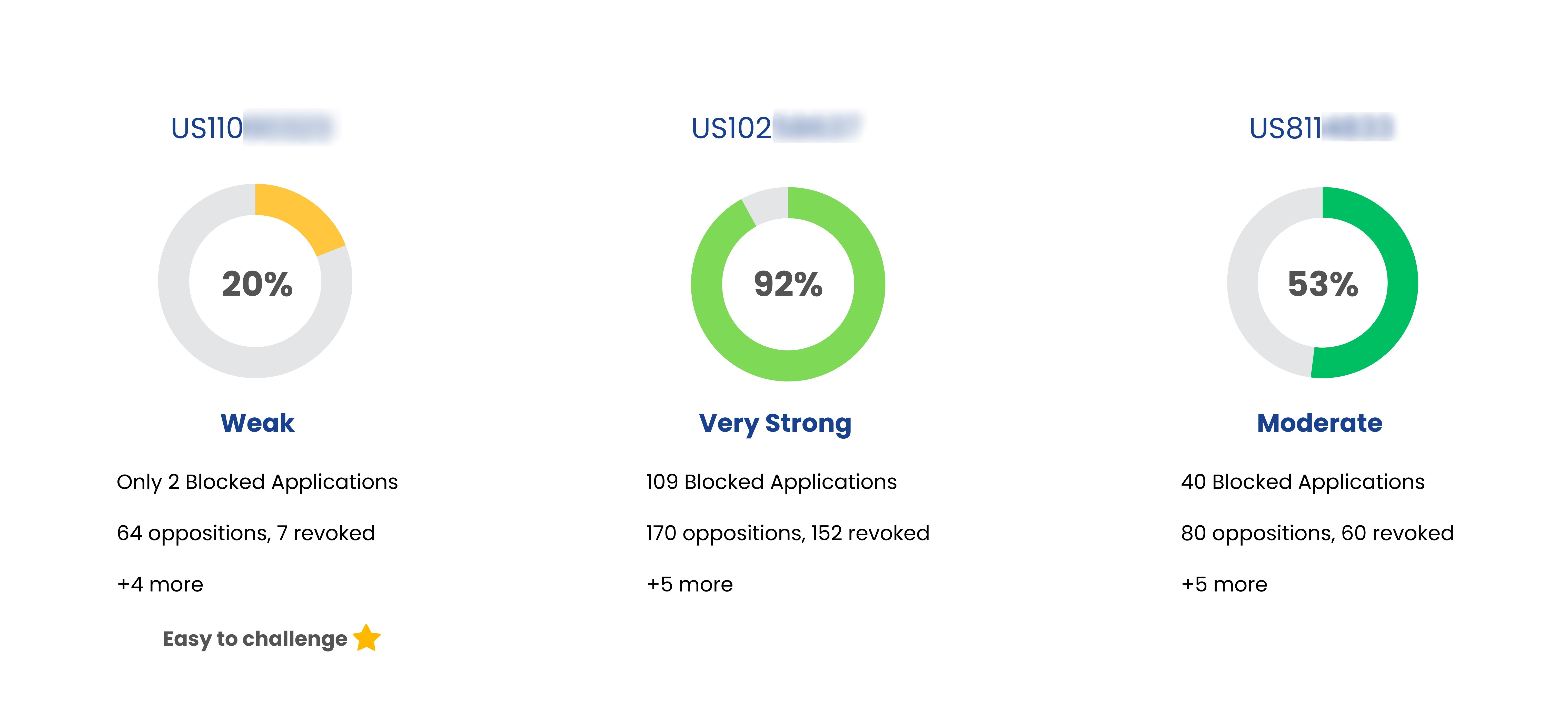
Coming Soon
Patent Strength Analyzer
YesNo
Thank you for your response 🥳
