Amlodipine Besylate; Hydrochlorothiazide; Valsartan Patent Expiration
Amlodipine Besylate; Hydrochlorothiazide; Valsartan is Used for treating high blood pressure. It was first introduced by Novartis Pharmaceuticals Corp
Amlodipine Besylate; Hydrochlorothiazide; Valsartan Patents
Given below is the list of patents protecting Amlodipine Besylate; Hydrochlorothiazide; Valsartan, along with the drug name that holds that patent and the company name owning that drug.
| Drug Used in | Drug Patent Number | Drug Patent Title | Drug Patent Expiry | Drug Owner |
|---|---|---|---|---|
| Exforge Hct |
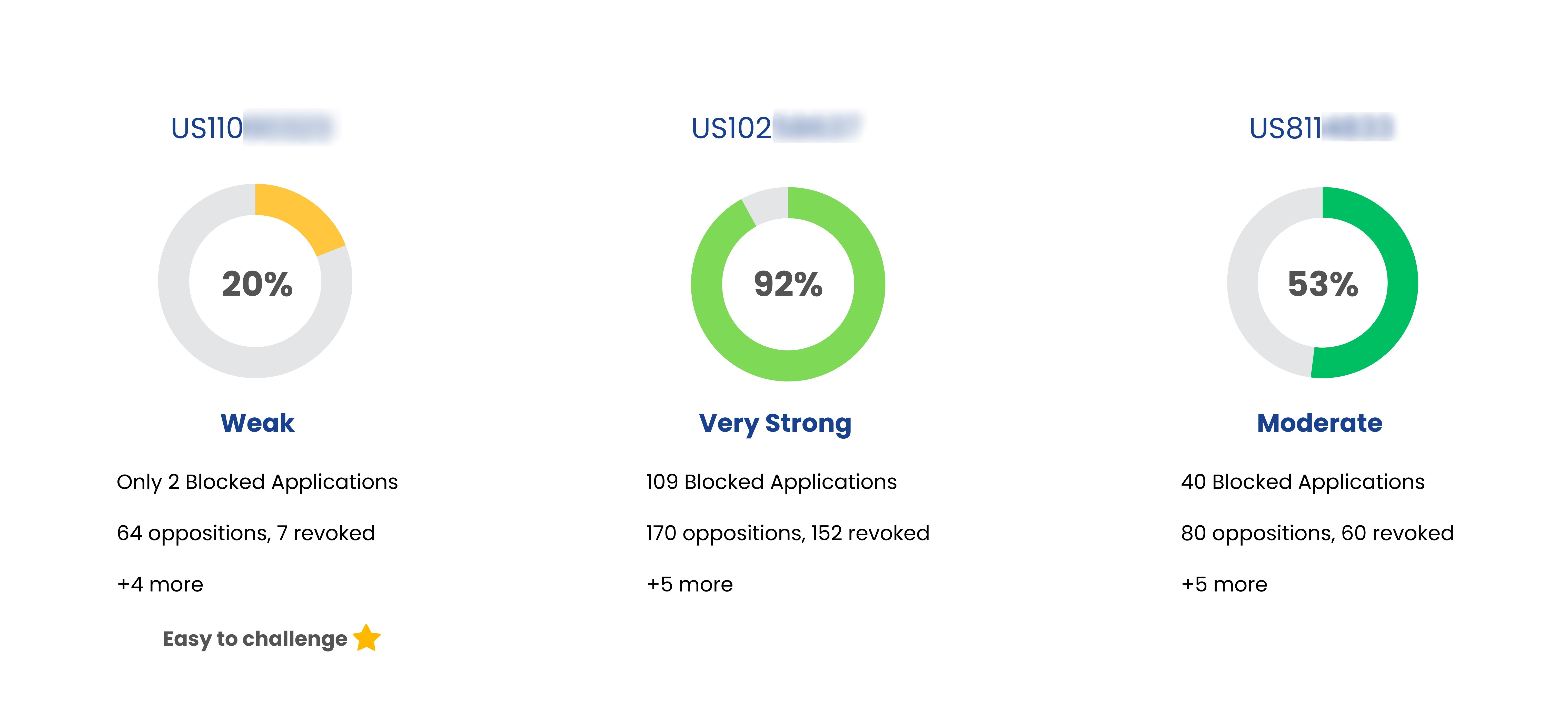
US5399578 (Pediatric) | Acyl compounds |
Sep 21, 2012
(Expired) | Novartis |
| Exforge Hct | US6294197 | Solid oral dosage forms of valsartan |
Jun 18, 2017
(Expired) | Novartis |
| Exforge Hct |
US6294197 (Pediatric) | Solid oral dosage forms of valsartan |
Dec 18, 2017
(Expired) | Novartis |
| Exforge Hct | US8101599 | Pharmaceutical composition containing anti-hypertensive agents |
May 16, 2023
(Expired) | Novartis |
| Exforge Hct | US8475839 | Solid dosage forms of valsartan, amlodipine and hydrochlorothiazide and method of making the same |
May 16, 2023
(Expired) | Novartis |
| Exforge Hct |
US8475839 (Pediatric) | Solid dosage forms of valsartan, amlodipine and hydrochlorothiazide and method of making the same |
Nov 16, 2023
(Expired) | Novartis |
Coming Soon
Patent Strength Analyzer
YesNo
Thank you for your response 🥳
Amlodipine Besylate; Hydrochlorothiazide; Valsartan Generics
Several generic applications have been filed for Amlodipine Besylate; Hydrochlorothiazide; Valsartan. The first generic version for Amlodipine Besylate; Hydrochlorothiazide; Valsartan was by Teva Pharmaceuticals Usa and was approved on Sep 25, 2012. And the latest generic version is by Aurobindo Pharma Ltd and was approved on Dec 19, 2017.
Given below is the list of companies who have filed for Amlodipine Besylate; Hydrochlorothiazide; Valsartan generic.
1. TORRENT
Torrent Pharmaceuticals Ltd has filed for 5 different strengths of generic version for Amlodipine Besylate; Hydrochlorothiazide; Valsartan. All of these versions come by the name AMLODIPINE BESYLATE, VALSARTAN AND HYDROCHLOROTHIAZIDE. Given below are the details of the strengths of this generic introduced by Torrent.
| Strength | Dosage Form | Availability | Application Pathway | TE code | Launch Date |
|---|---|---|---|---|---|
| EQ 10MG BASE; 25MG; 320MG | tablet | Discontinued | ORAL | N/A | Jun 3, 2015 |
| EQ 5MG BASE; 12.5MG; 160MG | tablet | Discontinued | ORAL | N/A | Jun 3, 2015 |
| EQ 5MG BASE; 25MG; 160MG | tablet | Discontinued | ORAL | N/A | Jun 3, 2015 |
| EQ 10MG BASE; 12.5MG; 160MG | tablet | Discontinued | ORAL | N/A | Jun 3, 2015 |
| EQ 10MG BASE; 25MG; 160MG | tablet | Discontinued | ORAL | N/A | Jun 3, 2015 |
2. LUPIN LTD
Lupin Ltd has filed for 5 different strengths of generic version for Amlodipine Besylate; Hydrochlorothiazide; Valsartan. All of these versions come by the name AMLODIPINE BESYLATE, VALSARTAN AND HYDROCHLOROTHIAZIDE. Given below are the details of the strengths of this generic introduced by Lupin Ltd.
| Strength | Dosage Form | Availability | Application Pathway | TE code | Launch Date |
|---|---|---|---|---|---|
| EQ 10MG BASE; 25MG; 320MG | tablet | Prescription | ORAL | AB | Jun 3, 2015 |
| EQ 5MG BASE; 25MG; 160MG | tablet | Prescription | ORAL | AB | Jun 3, 2015 |
| EQ 10MG BASE; 12.5MG; 160MG | tablet | Prescription | ORAL | AB | Jun 3, 2015 |
| EQ 10MG BASE; 25MG; 160MG | tablet | Prescription | ORAL | AB | Jun 3, 2015 |
| EQ 5MG BASE; 12.5MG; 160MG | tablet | Prescription | ORAL | AB | Jun 3, 2015 |
3. TEVA PHARMS
Teva Pharmaceuticals Usa has filed for 5 different strengths of generic version for Amlodipine Besylate; Hydrochlorothiazide; Valsartan. All of these versions come by the name AMLODIPINE BESYLATE, VALSARTAN AND HYDROCHLOROTHIAZIDE. Given below are the details of the strengths of this generic introduced by Teva Pharms.
| Strength | Dosage Form | Availability | Application Pathway | TE code | Launch Date |
|---|---|---|---|---|---|
| EQ 5MG BASE; 12.5MG; 160MG | tablet | Discontinued | ORAL | N/A | Sep 25, 2012 |
| EQ 5MG BASE; 25MG; 160MG | tablet | Discontinued | ORAL | N/A | Sep 25, 2012 |
| EQ 10MG BASE; 25MG; 160MG | tablet | Discontinued | ORAL | N/A | Sep 25, 2012 |
| EQ 10MG BASE; 25MG; 320MG | tablet | Discontinued | ORAL | N/A | Sep 25, 2012 |
| EQ 10MG BASE; 12.5MG; 160MG | tablet | Discontinued | ORAL | N/A | Sep 25, 2012 |
4. STRIDES PHARMA
Strides Pharma Global Pte Ltd has filed for 5 different strengths of generic version for Amlodipine Besylate; Hydrochlorothiazide; Valsartan. All of these versions come by the name AMLODIPINE BESYLATE, VALSARTAN AND HYDROCHLOROTHIAZIDE. Given below are the details of the strengths of this generic introduced by Strides Pharma.
| Strength | Dosage Form | Availability | Application Pathway | TE code | Launch Date |
|---|---|---|---|---|---|
| EQ 5MG BASE; 12.5MG; 160MG | tablet | Prescription | ORAL | AB | Jun 1, 2015 |
| EQ 5MG BASE; 25MG; 160MG | tablet | Prescription | ORAL | AB | Jun 1, 2015 |
| EQ 10MG BASE; 12.5MG; 160MG | tablet | Prescription | ORAL | AB | Jun 1, 2015 |
| EQ 10MG BASE; 25MG; 160MG | tablet | Prescription | ORAL | AB | Jun 1, 2015 |
| EQ 10MG BASE; 25MG; 320MG | tablet | Prescription | ORAL | AB | Jun 1, 2015 |
5. AUROBINDO PHARMA
Aurobindo Pharma Ltd has filed for 5 different strengths of generic version for Amlodipine Besylate; Hydrochlorothiazide; Valsartan. All of these versions come by the name AMLODIPINE BESYLATE, VALSARTAN AND HYDROCHLOROTHIAZIDE. Given below are the details of the strengths of this generic introduced by Aurobindo Pharma.
| Strength | Dosage Form | Availability | Application Pathway | TE code | Launch Date |
|---|---|---|---|---|---|
| EQ 5MG BASE; 25MG; 160MG | tablet | Prescription | ORAL | AB | Dec 19, 2017 |
| EQ 10MG BASE; 12.5MG; 160MG | tablet | Prescription | ORAL | AB | Dec 19, 2017 |
| EQ 10MG BASE; 25MG; 160MG | tablet | Prescription | ORAL | AB | Dec 19, 2017 |
| EQ 10MG BASE; 25MG; 320MG | tablet | Prescription | ORAL | AB | Dec 19, 2017 |
| EQ 5MG BASE; 12.5MG; 160MG | tablet | Prescription | ORAL | AB | Dec 19, 2017 |
